Invited presentations
Reality Capture for Disaster Recovery, Autodesk University, 11/16/17
Introducing GIS to contractors through UAS and GPS technology, ESRI IMF, 07/08/17
#045 – 3D ROBOTICS WITH DANIEL MCKINNON, Commercial Drones FM, 05/17/17
The Future of Flight, Harvard Business School Tech Conference, 10/15/2016
How Smart, Connected Products are Changing the World, GWC RobotX Meetup, 12/17/2015
Drones: Fun, Games, and Serious Business, SF Bar Association, 10/29/2015
From DIY to the Enterprise: The 3D Robotics Story, InterDrone, 09/11/2015
The Straight Story on UAVs in Agriculture, CITRIS Research Exchange Seminar Series, 09/01/2015
UAS Industry-Steps Toward Safe Integration Part I, NASA UTM 2015, 07/28/2015
UAVs in Agriculture, UC Davis Ag Innovation Entrepreneurship Academy, 04/23/2015
Panel II: Top Startups in the Drone Space, DroneComm 2015, 05/06/2015
UAVs in Agriculture: What do farmers really want?, DroneCon, 07/09/2014
Quoting articles
A product manager who worked at Meta and Google reveals which company is best for work-life balance
The Pragmatic Engineer Pulse #92, 05/02/24
State of the Drone Industry, Guinn Partners, 03/27/2018
The Evolution of 3DR – Commercial Drones FM Podcast Insights, Commerical UAV News, 05/31/2017
Flying High with Daniel McKinnon, VP Product, 3D Robotics, Autodesk blog, 05/09/2017
The Revolution of Drone-carried Sensors, GIM International, 03/31/2017
How Trump & Tech Will Affect Your Business in 2017, Construction Business Owner, 01/15/17
New rules for the sky, Construction Business Owner, 10/25/2016
Eye in the sky, Develop3D, 10/17/2016
3DR flies its Solo drone to new heights in bid for commercial business, Yahoo Tech, 07/22/2016
3DR Enterprise manager explains efforts for commercial markets, UAS Magazine, 07/21/2016
The Agribotix Guide to Drones in Agriculture, Sparkfun blog, 07/09/2014
How to Get the Most Out of Drone Images, AgWeb, 06/20/2014
Published academic articles
Gourab Kundu, Prahal Arora, Ferdi Adeputra, Polina Kuznetsova, Daniel McKinnon, Michelle Cheung, Larry Anazia, Geoffrey Zweig, “Multi-modal Content Localization in Videos Using Weak Supervision,” International Conference on Machine Learning (ICML), 2019. [PDF]
Identifying the temporal segments in a video that contain content relevant to a category or task is a difficult but interesting problem. This has applications in fine-grained video indexing and retrieval. Part of the difficulty in this problem comes from the lack of supervision since large-scale annotation of localized segments containing the content of interest is very expensive. In this paper, we propose to use the category assigned to an entire video as weak supervision to our model. Using such weak supervision, our model learns to do joint video level categorization and localization of content relevant to the category of the video. This can be thought of as providing both a classification label and an explanation in the form of the relevant regions of the video. Extensive experiments on a large scale data set show our model can achieve good localization performance without any direct supervision and can combine signals from multiple modalities like speech and vision.
D.D. McKinnon, “Process Extension from Embryonic Stem Cell-Derived Motor Neurons through Synthetic Extracellular Matrix Mimics,” ProQuest/UMI (2014). [PDF]
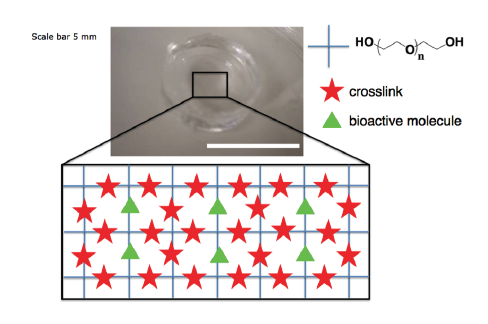 This thesis focuses on studying the extension of motor axons through synthetic poly(ethylene glycol) PEG hydrogels that have been modified with biochemical functionalities to render them more biologically relevant. Specifically, the research strategy is to encapsulate embryonic stem cell-derived motor neurons (ESMNs) in synthetic PEG hydrogels crosslinked through three different chemistries providing three mechanisms for dynamically tuning material properties. First, a covalently crosslinked, enzymatically degradable hydrogel is developed and exploited to study the biophysical dynamics of axon extension and matrix remodeling. It is demonstrated that dispersed motor neurons require a battery of adhesive peptides and growth factors to maintain viability and extend axons while those in contact with supportive neuroglial cells do not. Additionally, cell-degradable crosslinker peptides and a soft modulus mimicking that of the spinal cord are requirements for axon extension. However, because local degradation of the hydrogel results in a cellular environment significantly different than that of the bulk, enzymatically degradable peptide crosslinkers were replaced with reversible covalent hydrazone bonds to study the e↵ect of hydrogel modulus on axon extension. This material is characterized in detail and used to measure forces involved in axon extension. Finally, a hydrogel with photocleavable linkers incorporated into the network structure is exploited to explore motor axon response to physical channels. This system is used to direct the growth of motor axons towards co-cultured myotubes, resulting in the formation of an in vitro neural circuit.
This thesis focuses on studying the extension of motor axons through synthetic poly(ethylene glycol) PEG hydrogels that have been modified with biochemical functionalities to render them more biologically relevant. Specifically, the research strategy is to encapsulate embryonic stem cell-derived motor neurons (ESMNs) in synthetic PEG hydrogels crosslinked through three different chemistries providing three mechanisms for dynamically tuning material properties. First, a covalently crosslinked, enzymatically degradable hydrogel is developed and exploited to study the biophysical dynamics of axon extension and matrix remodeling. It is demonstrated that dispersed motor neurons require a battery of adhesive peptides and growth factors to maintain viability and extend axons while those in contact with supportive neuroglial cells do not. Additionally, cell-degradable crosslinker peptides and a soft modulus mimicking that of the spinal cord are requirements for axon extension. However, because local degradation of the hydrogel results in a cellular environment significantly different than that of the bulk, enzymatically degradable peptide crosslinkers were replaced with reversible covalent hydrazone bonds to study the e↵ect of hydrogel modulus on axon extension. This material is characterized in detail and used to measure forces involved in axon extension. Finally, a hydrogel with photocleavable linkers incorporated into the network structure is exploited to explore motor axon response to physical channels. This system is used to direct the growth of motor axons towards co-cultured myotubes, resulting in the formation of an in vitro neural circuit.
D.D. McKinnon, D.W. Domaille, T.E. Brown, K.A. Kyburz, E. Kiyotake, J.N. Cha, and K.S. Anseth, “Measuring Cellular Forces Using Bis-Aliphatic Hydrazone Crosslinked Stress-Relaxing Hydrogels,” Soft Matter (2014). [PDF]
Studies focused on understanding the role of matrix biophysical signals on cells, especially those when cells are encapsulated in hydrogels that are locally remodelled, are often complicated by appropriate methods to measure differences between the bulk and local material properties. From this perspective, stress-relaxing materials that allow long-term culture of embedded cells provide an opportunity to elucidate aspects of this biophysical signalling. In particular, rheological characterization of the stress relaxation properties allows one to link a bulk material measurement to local aspects of cellular functions by quantifying the corresponding cellular forces that must be applied locally. Here, embryonic stem cell-derived motor neurons were encapsulated in a well-characterized covalently adaptable bis-aliphatic hydrazone crosslinked PEG hydrogel, and neurite outgrowth was observed over time. Using fundamental physical relationships describing classical mechanics and viscoelastic materials, we calculated the forces and energies involved in neurite extension, the results of which provide insight to the role of biophysical cues on this process.
D.D. McKinnon,* T.E. Brown,* K.A. Kyburz, E. Kiyotake, and K.S. Anseth, “Design and Characterization of a Synthetically Accessible, Photodegradable Hydrogel for User-Directed Formation of Neural Networks,” Biomacromolecules (2014). [PDF]
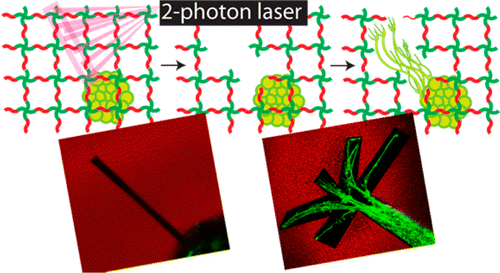 Hydrogels with photocleavable units incorporated into the cross-links have provided researchers with the ability to control mechanical properties temporally and study the role of matrix signaling on stem cell function and fate. With a growing interest in dynamically tunable cell culture systems, methods to synthesize photolabile hydrogels from simple precursors would facilitate broader accessibility. Here, a step-growth photodegradable poly(ethylene glycol) (PEG) hydrogel system cross-linked through a strain promoted alkyne–azide cycloaddition (SPAAC) reaction and degraded through the cleavage of a nitrobenzyl ether moiety integrated into the cross-links is developed from commercially available precursors in three straightforward synthetic steps with high yields (>95%). The network evolution and degradation properties are characterized in response to one- and two-photon irradiation. The PEG hydrogel is employed to encapsulate embryonic stem cell-derived motor neurons (ESMNs), and in situ degradation is exploited to gain three-dimensional control over the extension of motor axons using two-photon infrared light. Finally, ESMNs and their in vivo synaptic partners, myotubes, are coencapsulated, and the formation of user-directed neural networks is demonstrated.
Hydrogels with photocleavable units incorporated into the cross-links have provided researchers with the ability to control mechanical properties temporally and study the role of matrix signaling on stem cell function and fate. With a growing interest in dynamically tunable cell culture systems, methods to synthesize photolabile hydrogels from simple precursors would facilitate broader accessibility. Here, a step-growth photodegradable poly(ethylene glycol) (PEG) hydrogel system cross-linked through a strain promoted alkyne–azide cycloaddition (SPAAC) reaction and degraded through the cleavage of a nitrobenzyl ether moiety integrated into the cross-links is developed from commercially available precursors in three straightforward synthetic steps with high yields (>95%). The network evolution and degradation properties are characterized in response to one- and two-photon irradiation. The PEG hydrogel is employed to encapsulate embryonic stem cell-derived motor neurons (ESMNs), and in situ degradation is exploited to gain three-dimensional control over the extension of motor axons using two-photon infrared light. Finally, ESMNs and their in vivo synaptic partners, myotubes, are coencapsulated, and the formation of user-directed neural networks is demonstrated.
M.A. Azagarsamy,* D.D. McKinnon,* D.A. Alge, and K.S. Anseth, “Coumarin-Based Photodegradable Hydrogel: Design, Synthesis, Gelation, and Degradation Kinetics,” ACS Macro Letters (2014). [PDF]
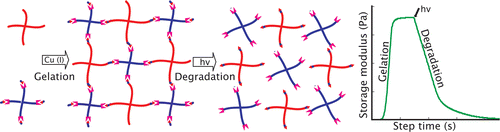 The design, synthesis, and characterization of a new class of coumarin-based photodegradable hydrogels are reported. Hydrogel formation was achieved rapidly and
The design, synthesis, and characterization of a new class of coumarin-based photodegradable hydrogels are reported. Hydrogel formation was achieved rapidly and
efficiently under aqueous conditions using copper-catalyzed click chemistry, which afforded excellent control over the rate of network formation. Rapid photodegradation, to the point of reverse gelation, was observed using both 365 and 405 nm light, and micrometer-scale features were eroded using two-photon irradiation at wavelengths as long as 860 nm.
D.D. McKinnon,* D.W. Domaille, J.N. Cha, and K.S. Anseth, “Bis-Aliphatic Hydrazone-Linked Hydrogels Form Most Rapidly at Physiological pH: Identifying the Origin of Hydrogel Properties with Small Molecule Kinetic Studies,” Chemistry of Materials (2014). [PDF]
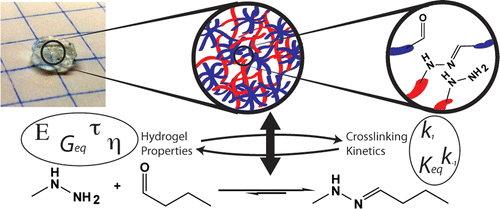 Rheological and small molecule kinetic studies were performed to study the formation and hydrolysis of the bis-aliphatic hydrazone bond. The rate of gelation was found to correspond closely with the rate of bond formation and the rate of gel relaxation with the rate of hydrolysis, indicating that small molecule kinetic studies can play an important role in material design. Furthermore, unlike aryl or acyl hydrazone bonds, the bis-aliphatic hydrazone bond forms rapidly under physiological conditions without requiring aniline catalysis yet maintains a pH-dependent rate of hydrolysis. These results suggest the bis-aliphatic hydrazone bond should find use alongside existing bioorthogonal click chemistries for bioconjugation, biomaterial synthesis, and controlled release applications.
Rheological and small molecule kinetic studies were performed to study the formation and hydrolysis of the bis-aliphatic hydrazone bond. The rate of gelation was found to correspond closely with the rate of bond formation and the rate of gel relaxation with the rate of hydrolysis, indicating that small molecule kinetic studies can play an important role in material design. Furthermore, unlike aryl or acyl hydrazone bonds, the bis-aliphatic hydrazone bond forms rapidly under physiological conditions without requiring aniline catalysis yet maintains a pH-dependent rate of hydrolysis. These results suggest the bis-aliphatic hydrazone bond should find use alongside existing bioorthogonal click chemistries for bioconjugation, biomaterial synthesis, and controlled release applications.
D.D. McKinnon, D.W. Domaille, J.N. Cha, and K.S. Anseth, “Biophysically Defined and Cytocompatible Covalently Adaptable Networks as Viscoelastic 3D Cell Culture Systems,” Advanced Materials (2013). [PDF]
Covalently crosslinked synthetic hydrogels are especially suitable as tissue engineering scaffolds due to their well-defined and easily tunable biochemical and biophysical properties. In order to enable complex cell functions like ECM deposition, motility, and spreading, a mechanism for crosslink degradation must be engineered into the material; however, the presence of a degradation trigger can complicate the cellular biophysical microenvironment. Furthermore, covalently crosslinked polymers typically produce a predominantly elastic material, while native tissues are complex viscoelastic structures. Here, we present a step-growth poly(ethylene glycol) (PEG) hydrogel crosslinked by reversible hydrazone bonds. The macromer components are readily synthesized from commercially available precursors, and the resulting gels form rapidly under physiological conditions and provide a non-toxic matrix that is suitable for cell culture. This material is capable of mimicking aspects of the viscoelastic properties of native tissues, and the dynamic stress relaxing crosslinks permit complex cellular functions to occur while retaining the benefits of traditional covalently crosslinked hydrogels. Taken together, these attributes make hydrazone crosslinked hydrogels a unique tool for designing viscoelastic scaffolds and studying cellular responses to scaffold elasticity.
D.D. McKinnon, A.M Kloxin, and K.S. Anseth, “Synthetic hydrogel platform for three-dimensional culture of embryonic stem cell-derived motor neurons,” Biomaterials Science (2013). [PDF]
Culturing mammalian neurons in three-dimensional (3D) microenvironments that more closely recapitulate critical biochemical and biophysical aspects of the developing or adult central nervous system (CNS) milieu remains a significant challenge in neurobiological studies and in regenerative medicine. Here, we aimed to exploit recent advances in poly(ethylene glycol) (PEG) hydrogel chemistries to define a synthetic niche capable of supporting the culture and axonal outgrowth of both aggregated and dissociated mouse embryonic stem cell-derived motor neurons (ESMNs). Using thiol-ene click chemistry to create peptide crosslinked PEG hydrogels, we identified a hydrogel formulation that promotes neuronal survival and axon outgrowth through cell-extracellular matrix interactions, such as those between the laminin-derived YIGSR and its integrin, and that allows neurons to remodel their extracellular environment through matrix metalloproteinase (MMP)-mediated polymer network degradation. Our results demonstrate a 3D platform for culture of both aggregated and single mammalian motor nerve cells that not only permits cell survival over more than a week of culture, but also allows for the robust extension of motor axons. In addition, the optical transparency of the hydrogel allows simultaneous imaging of live cell functions, and as such, this material system should prove useful for studying fundamental aspects of neuronal development.